Explore our lineup.
Black
Eko CORE™ Digital Attachment
Attachment that transforms your analog stethoscope to digital with amplification and noise cancellation.
$259
$224
SHOP NOW
Black
High Polish Rainbow
High Polish Copper
High Polish Mirror
3M™ Littmann® CORE Digital Stethoscope
Littmann® cardiology-grade stethoscope enhanced by Eko's CORE™ digital technology.
$379
$329
SHOP NOW
Silver
Matte Black
Champagne
Amethyst
Wintergreen
Sapphire
Electric Pink
Ruby Red
Cool Blue
Rose Gold
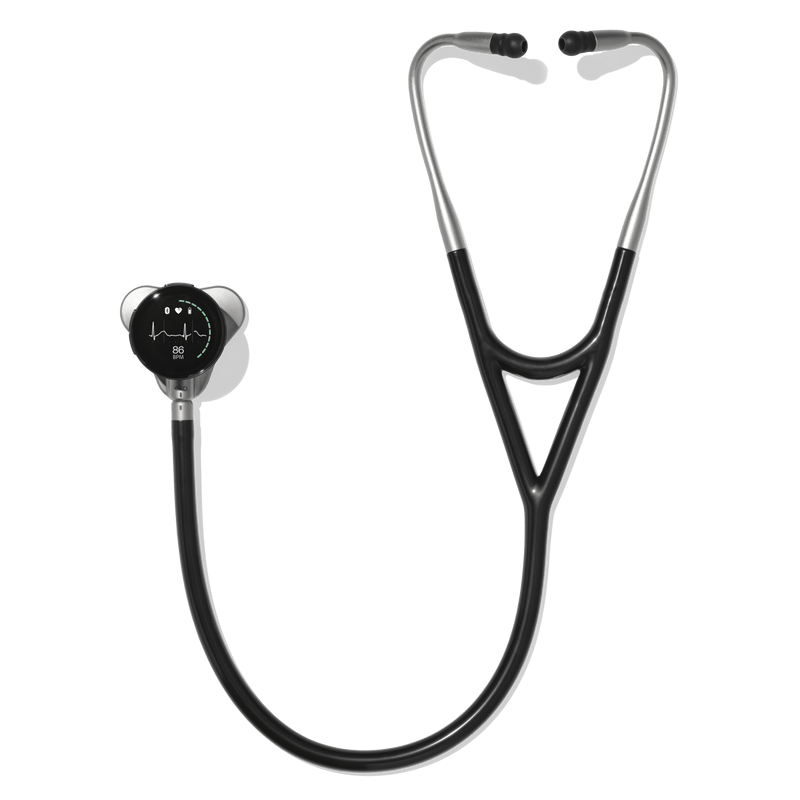
Eko CORE 500™ Digital Stethoscope
The modern stethoscope replacement, blending 3-lead ECG with AI insights like AFib and structural murmur detection.
$449
$399
SHOP NOW
Buy with confidence.
Accessorize your stethoscope.
MKT-0003672